Chronic Stress in Rats Impacts Astrocytic Number and Morphology in the Amygdala but Not in the Hippocampus
Dr. Rajbhandari is currently Assistant Reseach Professor at University of California, Los Angeles. Follow them on Twitter @CosmicOmBrain
Astrocytes are an abundant glial cell type in the brain and are involved in both healthy brain function and disease states. Studies have shown that astrocytic and neuronal functions are tightly linked. For example, morphological changes in astrocytes can alter neuronal functions including synaptic plasticity. Prior research has shown that chronic stress differentially alters neuronal morphology in the hippocampus and amygdala, two brain regions important for emotional regulation. However, less attention has been paid to the effects of stress on astrocytic morphology in these structures. In their eNeuro publication, Naskar and Chattarji found that chronic stress in rats alters the number and morphology of astrocytes in the basal nucleus of the amygdala (BA), but not in the CA3 region of the dorsal hippocampus (dHPC).
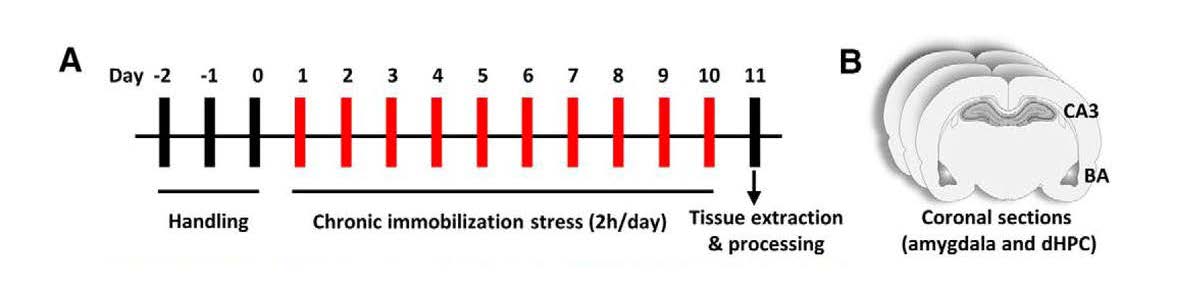
The authors subjected 2-month-old rats to a chronic stress immobilization protocol. Starting on Day 1, rats were completely immobilized in plastic immobilization bags for 2 hours a day for ten consecutive days (Figure 1). The rats were able to breathe through a hole in the bags adjacent to their nose but did not have access to food or water during the immobilization period. Control rats were not subjected to stress and were housed separately. On Day 11, brains were harvested, and coronal sections containing both the CA3 and BA were prepared for analysis of astrocyte number and morphology (Figure 1).
Figure 1. Schematic illustration of the experimental protocol. (A) After three days of handling, rats were subjected to chronic immobilization stress for 2 h per day for ten days. (B) On Day 11, brains were harvested, and coronal sections containing both CA3 and BA were used for immunofluorescence or morphometric analysis. (Adapted from Figure 1 in
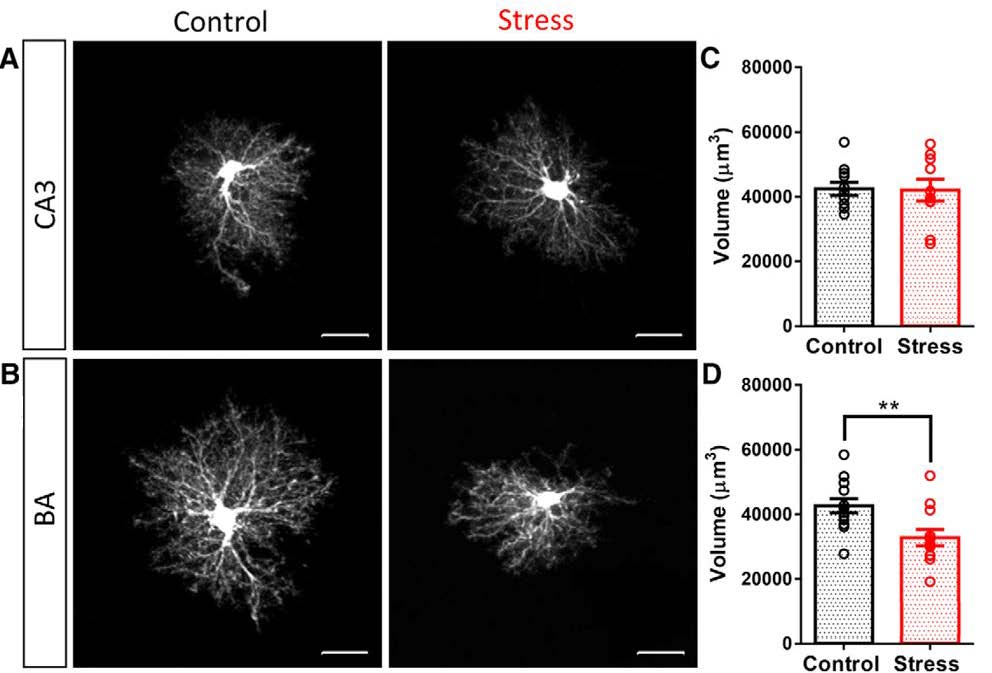
Expression of glial fibrillary acidic protein (GFAP), an intermediate filament protein expressed in astrocytes, revealed that after ten consecutive days of chronic immobilization stress, astrocyte number was decreased in the BA, but was unaffected in CA3. As GFAP immunoreactivity does not visualize the entirety of astrocyte morphology, individual astrocytes in coronal sections were filled with a fluorescent dye. This morphometric analysis showed that a reduction in the volume of astrocytic protrusions, in turn, caused a reduction in the volume of neuropil occupied by astrocytes in the BA (Figure 2). Neither effect was observed for CA3 astrocytes (Figure 2). The authors also verified that chronic immobilization stress resulted in dendritic hypertrophy in amygdalar neurons, corroborating findings from previous studies and validating their experimental approach. Notably, BA neurons exhibiting dendritic hypertrophy were located adjacent to astrocytes that had undergone atrophy, thereby providing striking examples of the opposite effects of the same stress on neurons versus astrocytes.
Figure 2. Chronic immobilization stress causes a reduction in the neuropil volume occupied by astrocytes in BA but not in CA3. (A,B) Representative images of dye-filled astrocytes in CA3 (A) and BA (B) in control rats (A,B, left) and rats subjected to chronic immobilization stress (A,B, right). (C,D) Effect of stress on neuropil volume of astrocytes in CA3 (C) and BA (D) in control rats (black) and rats subjected to stress (red). ** indicates p < 0.01 compared to control; circles represent data from individual rats. (Adapted from Figure 2 in
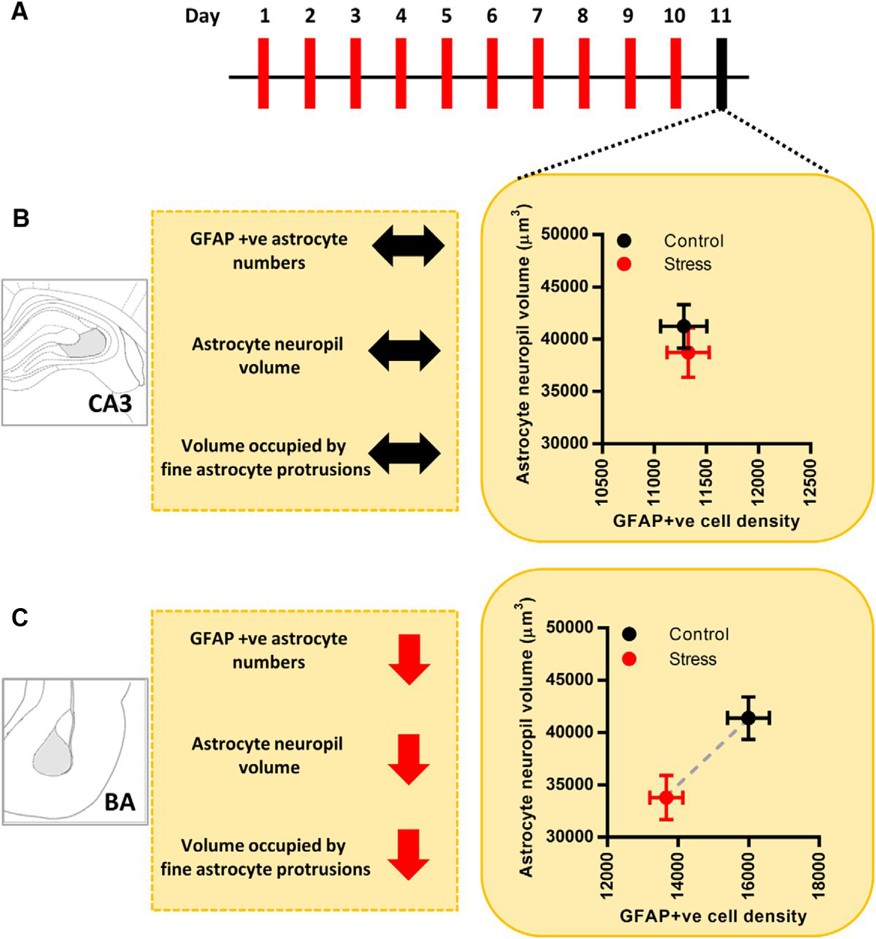
The overall findings of this study (Figure 3) highlight the importance of studying astrocytes in various brain areas after stress or any other behavioral manipulation. This study is important and timely as most research on stress effects focuses on neuronal rather than astrocytic function. However, understanding mechanisms that alter astrocyte number and morphology could lead to the development of novel or better therapeutic approaches for treating stress- and anxiety-related disorders. Future studies are needed to determine to what extent changes in astrocyte number and morphology are altered in neuropathological states. The eNeuro publication by Naskar and Chattarji is an important starting point for teasing apart the mechanisms that underlie the different effects that chronic stress can have on various cell types and brain regions.
Figure 3. Schematic illustration of the experimental protocol and summary of main findings. (A) Timeline of chronic immobilization stress and brain harvesting. (B) Effects of chronic immobilization stress on astrocytes in CA3. Astrocyte neuropil volume is represented along the y-axis, and GFAP-positive astrocyte numbers are represented along the x-axis. Stress leads to no significant changes in either the neuropil volume or the number of astrocytes in CA3 (control, black; stress, red). Stress leads to no change in the volume occupied by the fine protrusions of CA3 astrocytes. (C) Effects of chronic immobilization stress on astrocytes in BA. Astrocyte neuropil volume is represented along the y-axis, and GFAP-positive astrocyte numbers are represented along the x-axis. Stress leads to a significant reduction in both the neuropil volume and the number of astrocytes in BA (control, black; stress, red). Stress also leads to a significant reduction in the volume occupied by the fine protrusions of BA astrocytes; ↔ indicates no change; ↓ indicates decrease; - - - - indicates the difference between mean values. (Adapted from Figure 7 in
This Reader's Pick was reviewed and edited by eNeuro Features Editor Rosalind S.E. Carney, D.Phil.
Read the full article:
Stress Elicits Contrasting Effects on the Structure and Number of Astrocytes in the Amygdala versus HippocampusSaptarnab Naskar and Sumantra Chattarji
FOLLOW US
POPULAR POSTS
TAGS
CATEGORIES


 RSS Feed
RSS Feed




